Funding the Future of Bone Health: Scaling Osteoboost and Expanding Access
When a woman is diagnosed with osteopenia, there isn’t much to offer her. She’s often told to take calcium and vitamin D, exercise, and to come back when it gets worse. For decades, that has largely been the extent of it. Little innovation from big pharma (no new agents in clinical trials). No prescription devices. Just a condition affecting more than 44 million Americans, most of them women, quietly progressing without effective intervention. And what happens after progressions to Osteoporosis - only 10% of patients use the pharmaceutical agents.
That approach stands in sharp contrast to how most chronic conditions are managed today. Across much of medicine, the focus has shifted toward identifying risk earlier and intervening before the consequences become more serious. Bone health has lagged behind. Too often, low bone density is monitored rather than actively managed, leaving women in an under-addressed gap between diagnosis and fracture.
Today, we’re announcing $8 million in new funding to scale Osteoboost and change the standard of care for low bone density.
Backed against the odds
You’d think that kind of unmet need would be a magnet for investment. It hasn’t been.
Women’s health and medical devices both sit near the bottom of most funding charts. We are a female-led, non-AI, hardware company operating in one of healthcare’s most underfunded categories.
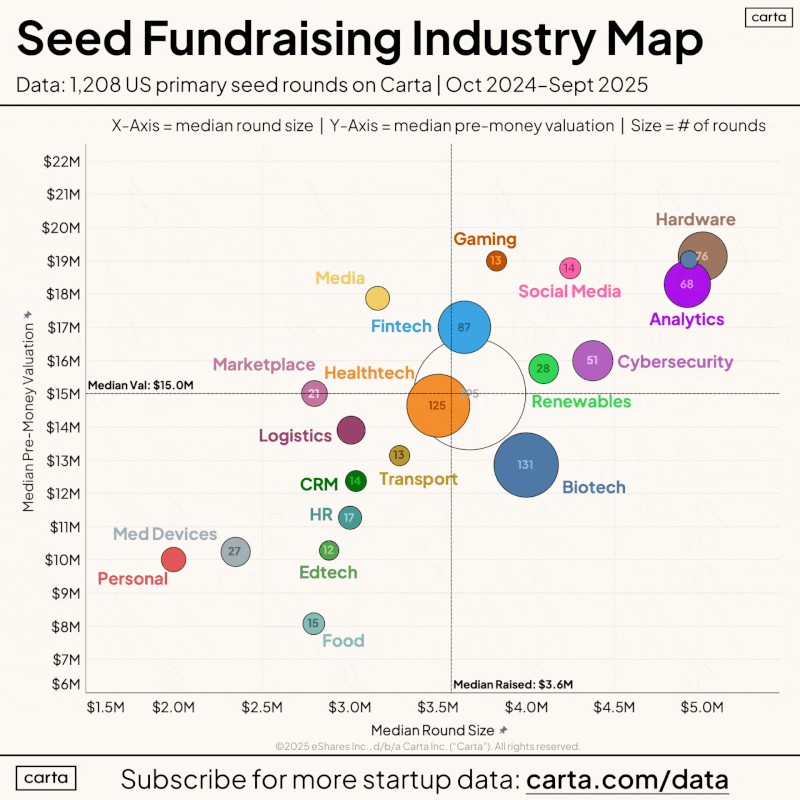
A recent Carta analysis of 1,209 seed rounds found that AI software companies raised at a $20 million median pre-money valuation. Hardware and medical devices ranked near the bottom. Women’s health receives just 6% of private healthcare capital, according to a January 2026 World Economic Forum report, with most of that focused on reproductive health and oncology.
We are also a regulated medical device company. Not software. Not AI. By most venture metrics, this is a harder category to fund (though one where it’s easier to protect IP). I knew this going in. What I didn’t fully anticipate was how clarifying it would be. When the market isn’t set up for you, the only reason to keep going is absolute conviction that the need is real and that the business opportunity is real. For Osteoboost, it is.
Why we kept going
Nearly 10 million Americans have osteoporosis. Another 44 million live with low bone density. For postmenopausal women especially, the window between diagnosis and fracture is long, silent, and largely unaddressed.
There are no new pharmaceutical agents in clinical trials for osteopenia, big pharma has exited seeing “low demand”, and until Osteoboost, there were no FDA-cleared prescription devices.
I came to this company because I was diagnosed with osteopenia myself, despite regular exercise and hitting my calcium goals. My doctor told me to keep doing what I was already doing. That was the entire treatment plan. I knew that couldn’t be the best we could offer women.
When neither the standard of care nor the outcomes have meaningfully changed for tens of millions of people, the decision to keep going isn't complicated.
Patient demand is what makes the case
Since we began shipping devices last May, more than 2,500 physicians have prescribed Osteoboost, including clinicians at more than 30 leading academic medical centers. We were also named to TIME’s 2025 Best Inventions list.
Patients aren’t waiting for the market to catch up. Low utilization rates of medication does not equal low concern or care about bone health.
What I hear most often from the women using Osteoboost is that they weren’t looking for a medical device. They were looking for something with clinical proven efficacy that matched the life they’re still living. They’re traveling. They’re exercising. They’re carrying grandchildren. Then they receive a diagnosis that quietly changes how they think about all of it.
Osteoboost gives them a way to act on that, not just live with it.
What this funding makes possible
This round, led by Ambit Health Ventures with new investors Emmeline Ventures, Disrupt Health Impact Fund, and others, will fund three priorities: scaling manufacturing to meet growing demand, expanding our clinical research program, and broadening our commercial reach to connect more women and physicians with Osteoboost.
It also moves us closer to something we've always believed: bone health requires more than a single intervention. That's why we built Wellen by Osteoboost, a physical therapist-developed training platform designed to work alongside the device. Users improved lower-body strength by 22.8% after one 18-session series, and nearly 80% of those who began at elevated fall risk improved to average or above-average levels.
A word on persistence
There is a generation of women who spent their careers pushing for equal opportunity and are now asking their doctors a different question: why aren’t there better options for me? Osteoboost exists because that question deserves a real answer. We intend to keep providing one.
To everyone who has prescribed Osteoboost, used it, advocated for it, or backed it: thank you.
To learn more or get started with a prescription, visit osteoboost.com.
References
Ready to get stronger?






